What is Down syndrome?
|
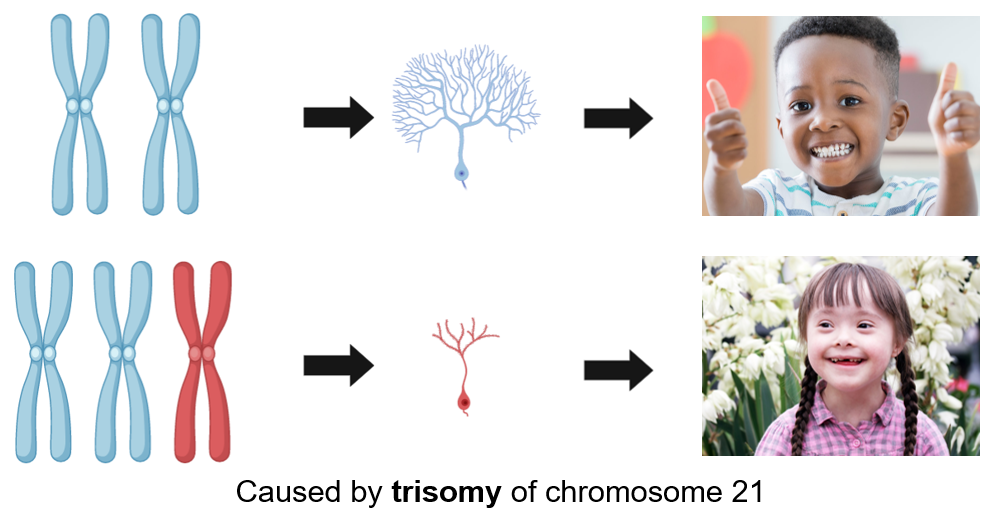
At an incidence of about 1 case per 800 live births, Down syndrome is the most common genetic development disorder and is caused by trisomy of chromosome 21. Down syndrome can be detected 12 to 13 weeks into pregnancy and will cause several abnormalities in affected individuals throughout their developmental years.
Down syndrome individuals have very distinct physical features that include: a short neck, flattened face and nose, small mouth and ears, upward slanting eyes, and decreased muscle tone. Other symptoms that aren't readily noticeable include cognitive and behavioral problems such as: slow learning, delayed language and speech [2,3]. |
Although a single, standard treatment for Down syndrome does not exist, personalized care that focuses on the affected individual's intellectual and physical needs can be implemented. Treatment options could include: speech therapy, physical therapy, and specialized education programs. [1]
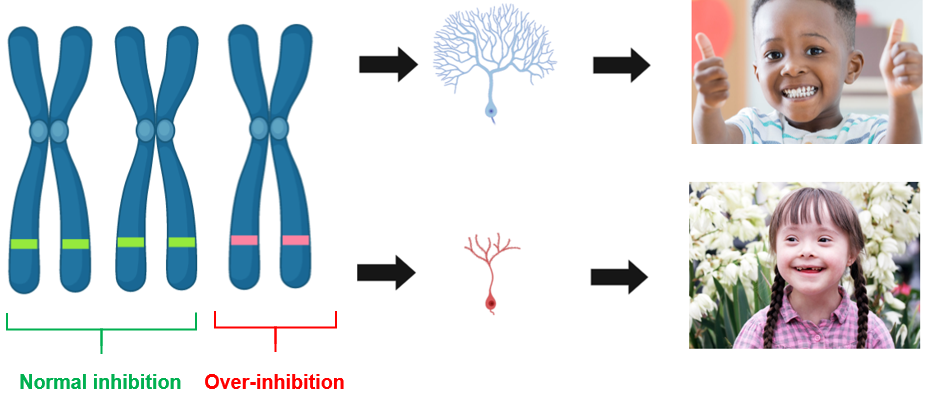
Chromosome 21 is known to have several genes that regulate neurogenesis and neural proliferation. When you have two copies of chromosome 21, you have normal neurogenesis and proliferation. Down syndrome patients have an extra chromosome, and this extra chromosome is linked to the decreased neurogenesis and proliferation observed in down syndrome patients.
Chromosome 21 is known to have several genes that regulate neurogenesis and neural proliferation. When you have two copies of chromosome 21, you have normal neurogenesis and proliferation. Down syndrome patients have an extra chromosome, and this extra chromosome is linked to the decreased neurogenesis and proliferation observed in down syndrome patients.
The relationship between DYRK1A and Down syndrome
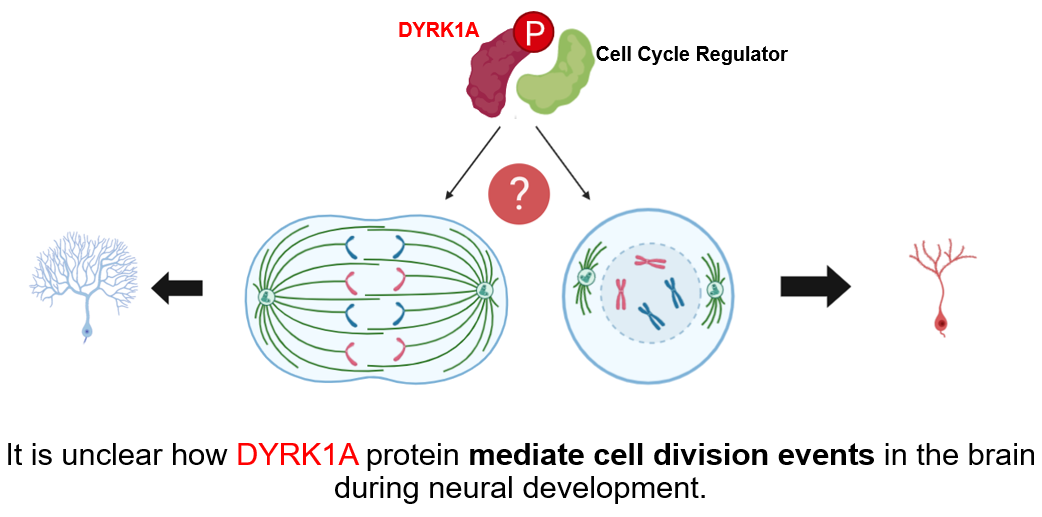
Chromosome 21 contains over 200 genes, but there have been several candidate genes identified that play a role in the cognitive impairment that occurs in individuals with Down syndrome. One of these candidate genes is DYRK1A, which encodes for the dual-specificity tyrosine-(Y)-phosphorylation-regulated kinase 1A protein. DYRK1A has received increased attention because it has been associated with cognitive defects in other disorders such as Alzheimer's disease. Because of the extra copy of chromosome 21, DYRK1A is overexpressed in both the fetal and adult periods. DYRK1A plays a role in reduced neurogenesis and premature neuronal differentiation of neuroprogenitor cells, so the overexpression of this gene can lead to several developmental deficits.
What is the gap in knowledge?
|
Although several genes may be involved, the DYRK1A is one gene that has been associated with cognitive defects in Down syndrome [2]. DYRK1A encodes a kinase that plays a role in neural development and neuronal differentiation [3]. Interestingly, DYRK1A also mediates cell proliferation [4], and it is unclear if the levels of DYRK1A protein may mediate cell division events during neural development. |
Zebrafish as a model organism
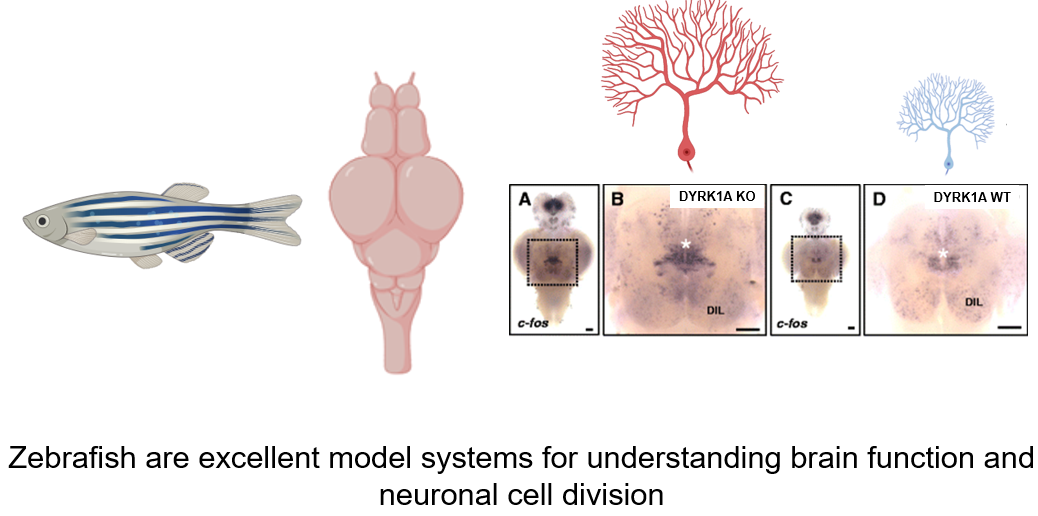
For the study of DYRK1A, and its role in the development of Down syndrome, zebrafish (Danio rerio) will be chosen as the model organism. Zebrafish will be an ideal model organism because the transparent nervous system allows for simple observation of neural cell proliferation. Additionally, the zebrafish nervous system is well-known to be very similar to the nervous system in humans. With further research in the DYRK1A protein homology, the protein domains present in humans have been found to be well conserved in zebrafish. Additionally, an important finding was that the interactions between DYRK1A and important cell cycle regulators in both humans and zebrafish are very similar. These findings make the zebrafish a suitable model to observe the connection between the levels of DYRK1A and neural cell proliferation.
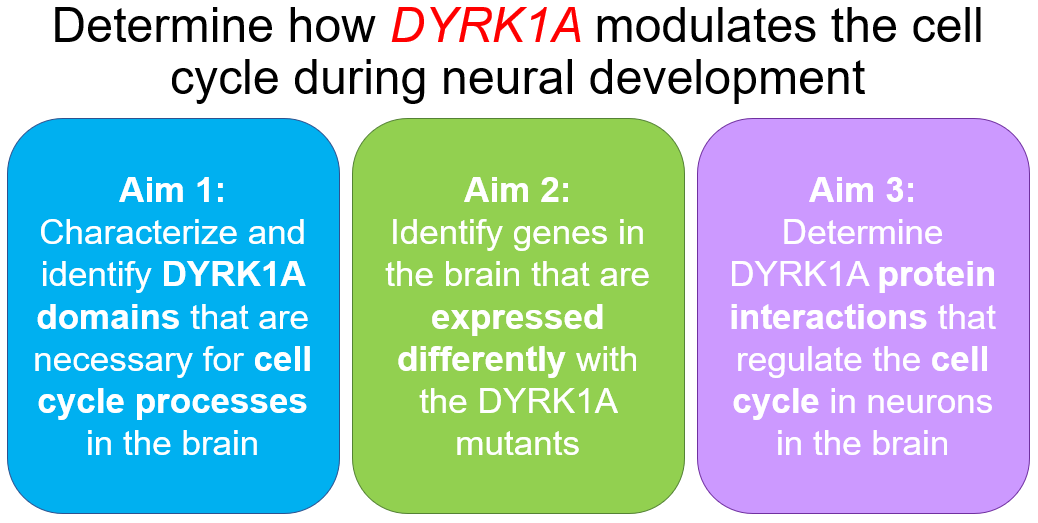
Specific Aims
Aim #1
|
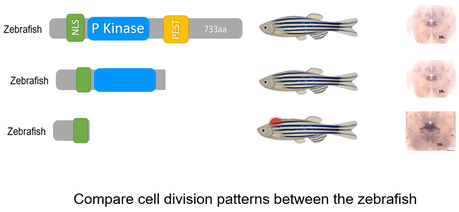
Aim 1: Identify the specific DYRK1A domains that are necessary for cell cycle processes in the brain.
Approach: First, NCBI BLAST will be used to determine the homologs of DYRK1A with diverse nervous systems. The homolog sequences will be aligned using ClustalOMEGA, and highly conserved domains will be determined. I will then create DYRK1A mutants by using CRISPR/Cas9 to knockout the identified domains one at a time. The neural proliferation in the mutant zebrafish will be compared with the neural proliferation in the wild-type zebrafish. Rationale: DYRK1A is known to reduce neuronal development and is also known to have phosphorylating capabilities through its P-kinase regions. Removing regions of the gene that may play a role in mediating the cell cycle in neurons is a priority as these will phenocopy the human disease. Hypothesis: Knockout of the P-kinase region in species with diverse nervous systems will be necessary for mediating cell division in neurons. |
Aim #2
|
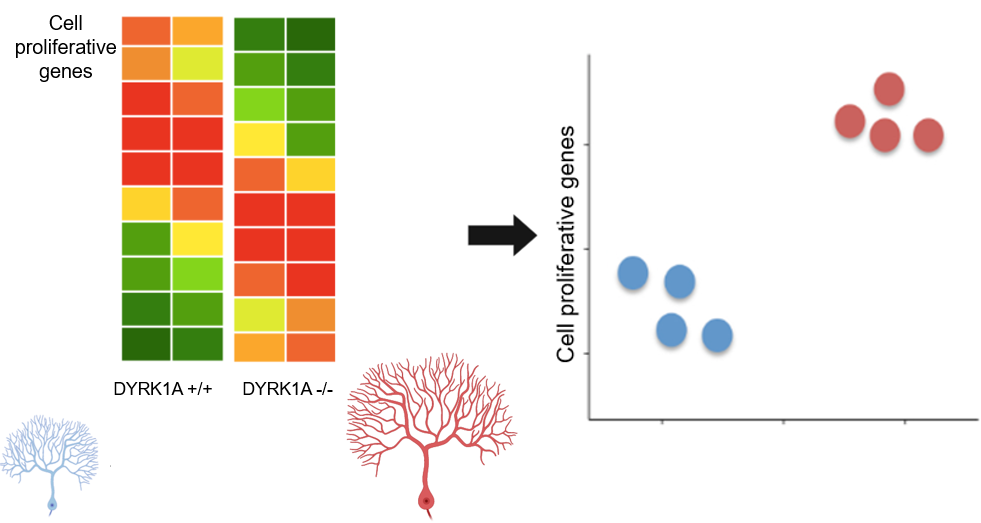
Aim 2: Identify genes in the brain that are expressed differently within the DYRK1A mutants
Approach: Brain tissue samples will be taken from both wild-type and the DYRK1A mutant zebrafish from Aim #1. The gene expression profiles of wild-type and P-kinase knockout mutants will be determined by conducting RNA sequencing on these tissue samples. Through GO analysis, the differentially expressed genes will be sorted into categories such as neurogenesis, neural cell differentiation, and neural cell proliferation. Rationale: Identifying genes that are differentially expressed in DYRK1A mutants will elucidate the interactions of DYRK1A that play a role in neural cell proliferation. Hypothesis: Wild-type and DYRK1A mutant zebrafish will differ in gene expression profiles related to neural cell proliferation. Specifically, the DYRK1A mutants without the P-kinase domain will have increased expression of neural cell proliferative genes. |
Aim #3
|
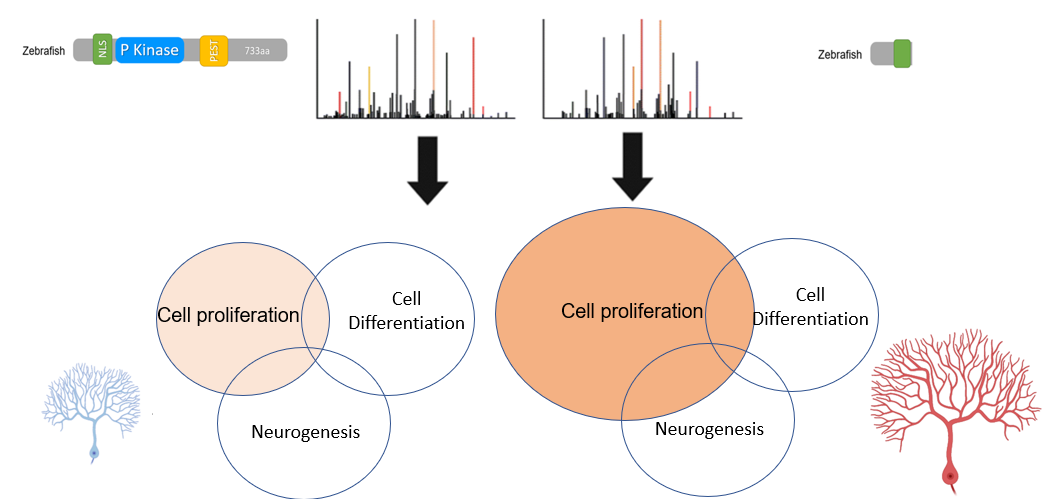
Aim 3: Determine DYRK1A protein interactions that regulate the cell cycle in neurons in the brain
Approach: I will determine the difference in protein interactions between DYRK1A protein with and without the P-kinase domain. By using BioID, proteins that interact with DYRK1A will be tagged in brain samples of wild-type and mutant zebrafish without a P-kinase domain. Using affinity chromatography, the proteins that are tagged from both samples will be purified and analyzed using mass spectrometry. Next, we can identify the proteins and group them by gene ontology groups such as neurogenesis, neural cell differentiation, and neural cell proliferation to understand the biological and functional relationships with DYRK1A. Rationale: Identifying DYRK1A protein interaction changes in the brain between wild-type and DYRK1A mutants will further elucidate the biological processes that mediate neural cell proliferation. Hypothesis: DYRK1A interacts with proteins that are implicated in mediating neural cell proliferation through its P-kinase domain. |
Through these specific aims, I expect to elucidate how the specific domains, gene expression profiles, and protein interactions of DYRK1A are involved in mediating cell division and neural proliferation in the brain. This research is especially crucial because it can lead to a better understanding of the biological mechanisms that lead to the pathology of neurodegenerative diseases such as Down syndrome.
Future Directions
In future studies, I would like to explore how the levels of DYRK1A directly affect learning and behavioral phenotypes in zebrafish. This would be especially interesting because Down syndrome is known to have several learning and behavioral impairments, and these studies could elucidate biological mechanisms that lead to these impairments as well as discover novel therapeutic routes for the neurodegenerative disorder.
| down_syndrome_rough_draft.pptx |
| mallela_4_5_2020.pptx |
| mallela_4_26_2020.pdf |
References
1. Feki, A., & Hibaoui, Y. (2018). DYRK1A Protein, A Promising Therapeutic Target to Improve Cognitive Deficits in Down Syndrome. Brain sciences, 8(10), 187. Retrieved from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6210095/
2. Nguyen, Thu Lan et al. “Correction of cognitive deficits in mouse models of Down syndrome by a pharmacological inhibitor of DYRK1A.” Disease models & mechanisms vol. 11,9 dmm035634. 27 Sep. 2018. Retrieved from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5125364/?report=classic
3. Liu, Y., Lin, Z., Liu, M., Wang, H., & Sun, H. (2017). Overexpression of DYRK1A, a Down Syndrome Candidate gene, Impairs Primordial Germ Cells Maintenance and Migration in zebrafish. Scientific reports, 7(1), 15313. Retrieved from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5681638
4. Thompson, B. J., Bhansali, R., Diebold, L., Cook, D. E., Stolzenburg, L., Casagrande, A. S., Besson, T., Leblond, B., Désiré, L., Malinge, S., & Crispino, J. D. (2015). DYRK1A controls the transition from proliferation to quiescence during lymphoid development by destabilizing Cyclin D3. The Journal of experimental medicine, 212(6), 953–970. https://doi.org/10.1084/jem.20150002
5. Olson L.E., et al. Down syndrome mouse models Ts65Dn, Ts1Cje, and Ms1Cje/Ts65Dn exhibit variable severity of cerebellar phenotypes. Dev. Dyn. 2004;230:581–589.
6. Liu, X, et al An AP-MS- and BioID-compatible MAC-tag enables comprehensive mapping of protein interactions and sub-cellular localizations. Nat Commun. 2018 Mar 22;9(1):1188. doi: 10.1038/s41467-018-03523-2.
7. Liu, X., Salokas, K., Tamene, F. et al. An AP-MS- and BioID-compatible MAC-tag enables comprehensive mapping of protein interactions and subcellular localizations. Nat Commun 9, 1188 (2018). https://doi.org/10.1038/s41467-018-03523-2
2. Nguyen, Thu Lan et al. “Correction of cognitive deficits in mouse models of Down syndrome by a pharmacological inhibitor of DYRK1A.” Disease models & mechanisms vol. 11,9 dmm035634. 27 Sep. 2018. Retrieved from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5125364/?report=classic
3. Liu, Y., Lin, Z., Liu, M., Wang, H., & Sun, H. (2017). Overexpression of DYRK1A, a Down Syndrome Candidate gene, Impairs Primordial Germ Cells Maintenance and Migration in zebrafish. Scientific reports, 7(1), 15313. Retrieved from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5681638
4. Thompson, B. J., Bhansali, R., Diebold, L., Cook, D. E., Stolzenburg, L., Casagrande, A. S., Besson, T., Leblond, B., Désiré, L., Malinge, S., & Crispino, J. D. (2015). DYRK1A controls the transition from proliferation to quiescence during lymphoid development by destabilizing Cyclin D3. The Journal of experimental medicine, 212(6), 953–970. https://doi.org/10.1084/jem.20150002
5. Olson L.E., et al. Down syndrome mouse models Ts65Dn, Ts1Cje, and Ms1Cje/Ts65Dn exhibit variable severity of cerebellar phenotypes. Dev. Dyn. 2004;230:581–589.
6. Liu, X, et al An AP-MS- and BioID-compatible MAC-tag enables comprehensive mapping of protein interactions and sub-cellular localizations. Nat Commun. 2018 Mar 22;9(1):1188. doi: 10.1038/s41467-018-03523-2.
7. Liu, X., Salokas, K., Tamene, F. et al. An AP-MS- and BioID-compatible MAC-tag enables comprehensive mapping of protein interactions and subcellular localizations. Nat Commun 9, 1188 (2018). https://doi.org/10.1038/s41467-018-03523-2
This web page was produced as an assignment for Genetics 564, an undergraduate capstone course at UW-Madison